Ziehl-Neelsen technique (AFB Staining): Principle, Procedure and reporting
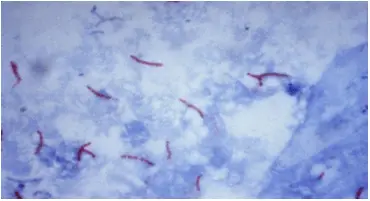
Ziehl-Neelsen (ZN) method of Acid Fast staining technique is used to stain Mycobacterium species including M. tuberculosis, M. ulcerans, and M. leprae and nontuberculous mycobacteria (NTM). Detection of acid-fast bacilli (AFB) in stained and acid-washed smears examined microscopically may provide the initial bacteriologic evidence of the presence of mycobacteria in a clinical specimen. Smear microscopy is the quickest and easiest procedure that can be performed.

The cellwall of Mycobacteria contain high concentration of lipid making them waxy, hydrophobic, and impermeable to routine stain such as the Gram Stain. They are also resistant to acid and alcohol and is described as Acid Fast Bacilli (AFB) or Acid Alcohol Fast Bacilli (AAFB).
There are two procedures commonly used for acid-fast staining:
- Carbolfuchsin methods which include the Ziehl-Neelsen and Kinyoun methods
( Light /bright field microscope) - Fluorochrome procedure using auramine-O or auramine-rhodamine dyes
(Fluorescent microscope).
Principle of Ziehl-Neelsen method of acid-fast staining
Mycobacteria, which do not stain well by Gram stain, are stained with carbol fuchsin combined with phenol.
- In the ‘hot’ ZN technique, the phenol-carbol fuchsin stain is heated to enable the dye to penetrate the waxy mycobacterial cell wall.
- In the ‘cold’ technique known as Kinyoun Method, stain are not heated but the penetration is achieved by increasing concentration of basic fuchsin and phenol and incorporating a ‘wetting agent’ chemical.
The stain binds to the mycolic acid in the mycobacterial cell wall. After staining, an acid decolorizing solution is applied. This removes the red dye from the background cells, tissue fibres, and any organisms in the smear except mycobacteria which retain (hold fast to) the dye and are therefore referred to as acid fast bacilli (AFB).
Following decolorization, sputum smear is counterstained with malachite green, or methylene blue which stains the background material, providing a contrast colour against which the red AFB can be seen.
Among the Mycobacterium species, M. tuberculosis and M. ulcerans are strongly acid fast. When staining specimens for these species, a 3% v/v acid alcohol is used to decolorize the smear, where as M. lepraeis only weakly acid fast. 0.5-1% v/v decolorizing solution is therefore used for M. leprae smears and also different staining and decolorizing time.
Note: 0.5% Acid alcohol or 5% Sulphuric acid is used for Atypical AFB because they (eg. Mycobacterium leprae, Nocardia asteroides) are much less acid and alcohol fast than Mycobacterium tuberculosis bacilli.
Sample Collection & Preparation : Due to overnight accumulation of secretions, first morning specimens are more likely to yield better recovery of AFB.
- Direct Smear: Smear prepared directly from a patient specimen prior to processing.
- Indirect Smear: Smear prepared from a processed specimen after centrifugation (is used to concentrate the material)
Reagents required:
- Carbol fuchsin stain (filtered)
- Acid alcohol 3% v/v (or 20% sulfuric acid)
- Malachite green 5 g/l (0.5% w/v) or Methylene blue, 5g/l
Ziehl-Neelsen Staining procedure
- Spread the sputum evenly over the central area of the slide using a continuous rotational movement. The recommended size of the smear is about 20 mm by 10 mm.
- Place slides on dryer with smeared surface upwards, and air dry for about 30 minutes.

Fig. Heat Fixation of smear (Upper: using electric heater, lower: using burner) - Heat fix dried smear.
- Cover the smear will carbol fuchsin stain
- Heat the smear until vapour just begins to rise (i.e. about 60 degree Celsius). Do not overheat (boil or dry). Add additional stain if necessary. Allow the heated stain to remain on the slide for 5 minutes.
- Wash off the stain with clean water.
- Cover the smear with 3% v/v acid alcohol for 2-5 minutes (or 20% sulfuric acid) or until the smear is sufficiently decolorized, i.e. pale pink.
Note: Check to see that no more red color runs off the surface when the slide is tipped. Add a bit more decolorizer for very thick slides or those that continue to “bleed” red dye. - Wash well with clean water
- Cover the stain with malachite green stain for 1-2 minutes
- Wash off stain with clean water
- Wipe the back of the slide clean, and place it in a draining rack for smear to air dry (DO NOT BOLT DRY).
- Examine the smear microscopically, using the 100x oil immersion objective (10X eye piece for a total of 1000X magnifi cation) and scan the smear systematically.
Procedural note:
- Heat fixation of untreated specimen may NOT kill M. tuberculosis (exercise care when handling slides) whereas alcohol fixation is bactericidal.
- Acid alcohol is flammable, therefore use it with care.
- Take great care while heating carbol fuchsin (as staining rack may contain flammable chemicals) to reduce the fire risk.
- Slides must not touch each other when placed on staining rack to prevent transfer of material
from one slide to another.
Results:
| Reagent | Acid Fast | Non-Acid Fast |
| Carbol Fuchsin with heat | Red (Hot Pink) | Red (Hot Pink) |
| Acid Alcohol | Red | Colorless |
| Methylene Blue/Malachite Green | Red | Blue/Green |
- AFB: Red, straight or slightly curved rods, occurring singly or in small groups, may appear beaded.
- Cells: Green
- Background material: Green
Reporting of sputum smear
- When no AFB are seen after examining 300 fields, report the smear as ‘No AFB seen’.
- When very few AFB are seen i.e. when only 1 or 2 AFB are seen after examining 100 fields, request a further specimen to examine (Those AFB might have came from tap water (saprophytic mycobacteria), or it may be scratch of glass slide or by the use of same piece of blotting paper while drying.
- When any red bacilli are seen, report the smear as ‘AFB positive’ and give an indication of the number of bacteria present as follows (The greater the number, the more infectious the patient):
- More than 10 AFB/field at least in 20 fields: report as + + +
- 1-10 AFB/field at least in 50 fields: report as + +
- 10-99 AFB/ 100 fields: report as +
- 1-9 AFB/100 fields: report the exact number
Limitation of AFB Microscopy
- Does not distinguish between viable and dead organisms
- Follow-up specimens from patients on treatment may be smear positive yet culture negative
- Limited sensitivity
- High bacterial load 5,000-10,000 AFB /mL is required for detection ( In contrast, 10 to 100 bacilli are needed for a positive culture).
- Many TB patients have negative AFB smears with a subsequent positive culture. Negative smears do not exclude TB disease.
- Limited specificity
- All mycobacteria are acid fast
- Does not provide species identification
- Local prevalence of MTB and NTM determine the predictive values of a positive smear for MTB
List of Acid Fast organisms (Other than Mycobacteria)
- Nocardia spp: Partial Acid Fast
- Rhodococcus spp: Partial Acid Fast
- Legionella micdadei: Partially acid fast in tissue
- Cyst of Cryptosporidium: Acid Fast
- Cyst of Isospora: Acid Fast