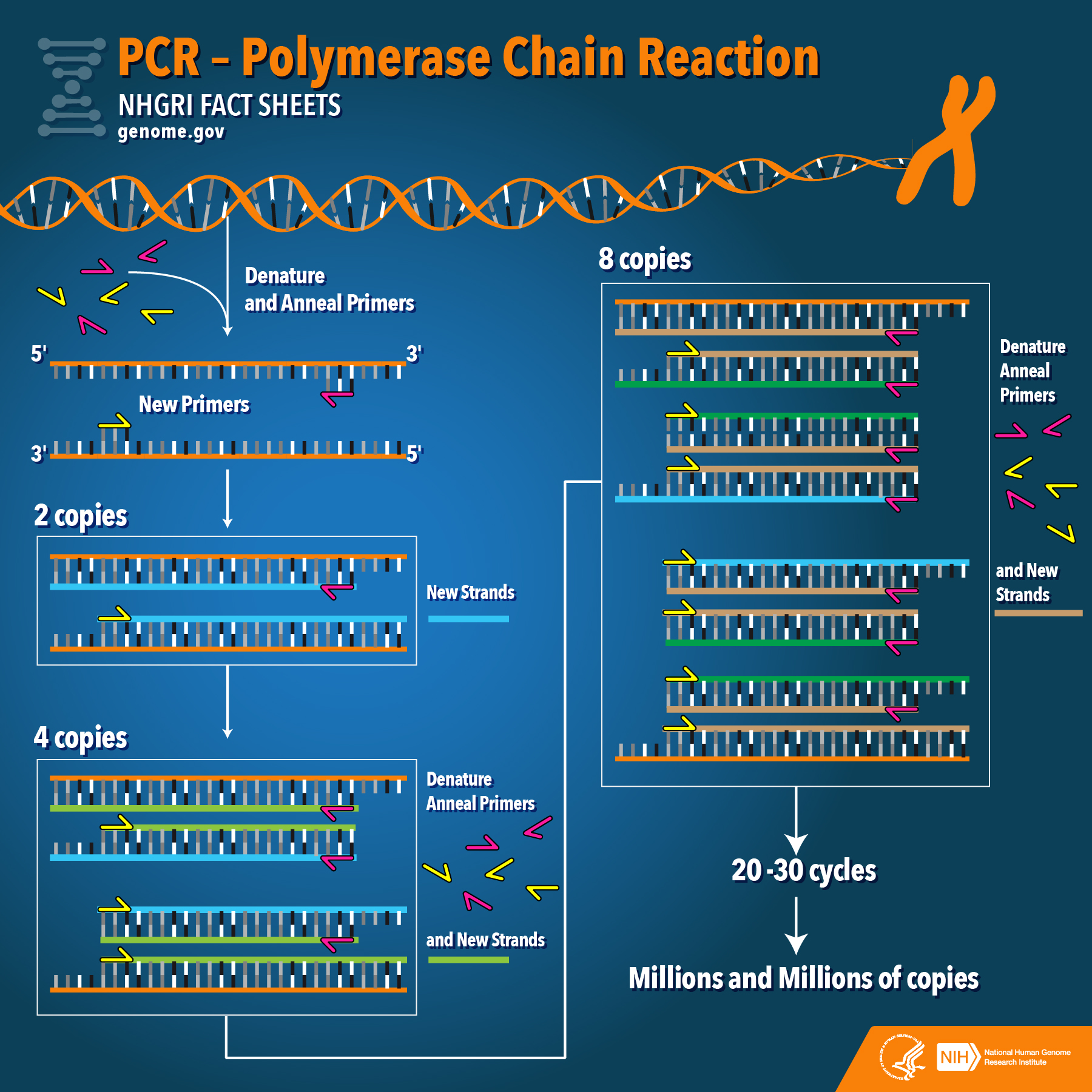
A thermal cycler (also known as a PCR machine or thermocycler) is a laboratory instrument that heats and cools samples in repetitive cycles to facilitate DNA or RNA amplification through the polymerase chain reaction. These machines have become commonplace in labs, although the introduction of mini or portable thermal cyclers has made it easier to perform PCR reactions in the field and classroom. Many thermal cyclers are programmable and allow users to save frequently used protocols. PCR typically requires 20 to 35 cycles comprising two to three temperature steps (denaturation, annealing, and extension).
Early PCR relied on a system of water baths. Thermal cyclers were introduced in the 1980s and became commercially available to researchers at the end of the decade. Over the years, the models have become smaller and lighter. Machines used for real-time quantitative PCR (qPCR) are still quite large, but they monitor the progression of the reaction in real time, eliminating the need for gel electrophoresis and UV gel documentation or imaging. Thermal cycler
The thermal cycler (also known as a thermocycler, PCR machine or DNA amplifier) is a laboratory apparatus most commonly used to amplify segments of DNA via the polymerase chain reaction (PCR).[1] Thermal cyclers may also be used in laboratories to facilitate other temperature-sensitive reactions, including restriction enzyme digestion or rapid diagnostics.[2] The device has a thermal block with holes where tubes holding the reaction mixtures can be inserted. The cycler then raises and lowers the temperature of the block in discrete, pre-programmed steps.
History[edit]
The earliest thermal cyclers were designed for use with the Klenow fragment of DNA polymerase I. Since this enzyme is destroyed during each heating step of the amplification process, new enzyme had to be added every cycle. This led to a cumbersome machine based on an automated pipettor, with open reaction tubes. Later, the PCR process was adapted to the use of thermostable DNA polymerase from Thermus aquaticus, which greatly simplified the design of the thermal cycler. While in some old machines the block is submerged in an oil bath to control temperature, in modern PCR machines a Peltier element is commonly used. Quality thermal cyclers often contain silver blocks to achieve fast temperature changes and uniform temperature throughout the block. Other cyclers have multiple blocks with high heat capacity, each of which is kept at a constant temperature, and the reaction tubes are moved between them by means of an automated process. Miniaturized thermal cyclers have been created in which the reaction mixture moves via channel through hot and cold zones on a microfluidic chip. Thermal cyclers designed for quantitative PCR have optical systems which enable fluorescence to be monitored during reaction cycling.
Modern innovations[edit]
Modern thermal cyclers are equipped with a heated lid that
presses against the lids of the reaction tubes. This prevents condensation of water from the reaction mixtures on the insides of the lids. Traditionally, a layer of mineral oil was used for this purpose. Some thermal cyclers are equipped with a fully adjustable heated lid to allow for nonstandard or diverse types of PCR plasticware.[3]
Some thermal cyclers are equipped with multiple blocks allowing several different PCR reactions to be carried out simultaneously. Some models also have a gradient function to allow for different temperatures in different parts of the block. This is particularly useful when testing suitable annealing temperatures for
PCR primers
PCR ThermocyclersDifferent types of thermocyclers available:Standard PCR Machines use interchangeable blocks to accommodate multi-well plates or tube strips, with a variety of program options for diverse gene amplification needs. Real-time themocyclers are used in applications such as quantitative gene expression analysis, SNP analysis, drug target validation and genotyping.Purchasing Considerations:When choosing a thermocycler, consider the types of applications needed, the sample number and volume, and options such as interchangeable blocks. Gradient options allow a range of temperatures across the block, useful for optimizing PCR conditions such as primer annealing temperatures. Multiple, smaller blocks at different temperatures also allow more flexibility. Also consider the capability to upgrade a standard machine to a real-time thermocycler, as well as space-saving footprints and touch-screen operation |
Modern innovations
Modern thermal cyclers are equipped with a heated lid that presses against the lids of the reaction tubes. This prevents condensation of water from the reaction mixtures on the insides of the lids. Traditionally, a layer of mineral oil was used for this purpose. Some thermal cyclers are equipped with a fully adjustable heated lid to allow for nonstandard or diverse types of PCR plasticware.[3]
Some thermal cyclers are equipped with multiple blocks allowing several different PCR reactions to be carried out simultaneously. Some models also have a gradient function to allow for different temperatures in different parts of the block. This is particularly useful when testing suitable annealing temperatures for PCR primers.
The Invention of PCR
 Few technical breakthroughs have changed the face of their field like the Polymerase Chain Reaction (PCR). Gene cloning, sequencing of complex genomes, DNA fingerprinting and DNA-based diagnostics are just some of the techniques that were either inefficient, crude or plain impossible before PCR. The technique has revolutionized biological research and biotechnology to such an extent that it can be considered as one of the major reasons for the boom the field has experienced over the last 20 years or so.
Few technical breakthroughs have changed the face of their field like the Polymerase Chain Reaction (PCR). Gene cloning, sequencing of complex genomes, DNA fingerprinting and DNA-based diagnostics are just some of the techniques that were either inefficient, crude or plain impossible before PCR. The technique has revolutionized biological research and biotechnology to such an extent that it can be considered as one of the major reasons for the boom the field has experienced over the last 20 years or so.
Kary Mullis is generally credited with inventing PCR in 1983 while working for Cetus Corporation in Emeryville, California. Mullis’ role at Cetus was to synthesise oligonucleotides for groups working on, amongst other things, methods to detect point mutations in human genes. Mullis was hatching an idea to detect the point mutations using Sanger-type DNA sequencing, employing DNA polymerase in the presence of an oligonucleotide primer and ddNTPs. The problem was that sequencing a single copy gene within the expanses of the human genome was impossible; the primer would bind in too many places. What he needed was a way to increase the concentration of the specific gene of interest.
While driving his Honda Civic on Highway 128 from San Francisco to Mendocino, Mullis made an intellectual leap. He reasoned that by using two opposed primers, one complementary to the upper strand and the other to the lower, then performing multiple cycles of denaturation, annealing and polymerization he could exponentially amplify the piece of DNA between the primers.
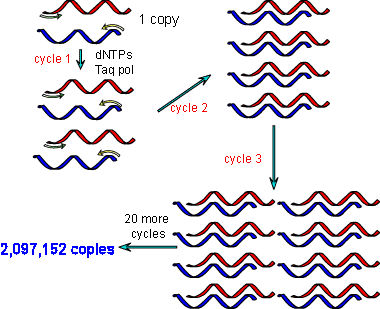
The idea of PCR was born, but the technique was still very much in it’s infancy. The E.coliDNA polymerase used in the early days was destroyed during the denaturation step so had to be replenished after every cycle. Cetus workers quickly developed the first thermal cycler named “Mr Cycle”, which automatically added new polymerase after each heating step.
In 1985, Mullis came up with the idea of using polymerase isolated from the extremophilic bacterium Thermophilus aquaticus. The polymerase, known as Taq polymerase, has optimal activity at 72°C and can withstand the 94°C required for denaturation of the DNA, meaning that many reaction cycles could be performed without being replenishing the enzyme. This breakthrough, together with advances in oligonucleotide synthesis made PCR both cost effective and convenient and it quickly entered mainstream research.
As researchers flocked to PCR, improvements to and variations on the process have, and continue to, come quickly and there are now hundreds of PCR-based applications in use in a variety of fields. Stephen Scharf, Mullis’ former colleague at Cetus, put it quite nicely:
One of PCR’s distinctive characteristics is unquestionably its extraordinary versatility. That versatility is more than its “applicability” to many different situations. PCR is a tool that has the power to create new situations for its use and those required to use it.
Perhaps the most influential of all techniques enabled by PCR is massive-scale genomic sequencing, which itself has transformed the biological and biotechnological research arena.
Mullis received the Nobel Prize for his ground-breaking invention in 1993. He also received a $10,000 bonus from his employers, Cetus, who later sold the patent rights to Hoffmann La-Roche for a cool $300,000,000. Seems to me there’s a lesson in there somewhere…
The Thermal Cycler (also known as a Thermocycler, PCR Machine or DNA Amplifier) is a laboratory apparatus used to amplify segments of DNA via the Polymerase Chain Reaction (PCR). The device has a thermal block with holes where tubes holding the PCR reaction mixtures can be inserted. The cycler then raises and lowers the temperature of the block in discrete, pre-programmed steps.
It is an important instrument which is needed in all the labs working in the field of gene cloning.
It is an important instrument which is needed in all the labs working in the field of gene cloning.
A basic PCR set up requires several components and reagents. These components include:
- DNA template that contains the DNA region (target) to be amplified.
- Two primers, which are complementary to the DNA regions at the 5' (five prime) or 3' (three prime) ends of the DNA region.
- A thermostable DNA polymerase such as Taq polymerase .
- Deoxynucleoside triphosphates (dNTPs), the building blocks from which the DNA polymerases synthesizes a new DNA strand.
- Buffer solution, providing a suitable chemical environment for optimum activity and stability of the DNA polymerase.
- Divalent cations, magnesium or manganese ions; generally Mg2+ is used Monovalent cation potassium ions.
Unique Features:
There are many companies making Thermocyclers such as BioRad, Thermo, Eppendorf, having the basic set-up with some specific features:
The Eppendorf Mastercycler personal is a high quality, compact thermal cycler. In addition to its speed and precise block homogeneity, it offers first-rate operational flexibility. Its universal block with a 25-well format can accommodate 25 x 0.2 ml tubes,16 x 0.5 ml tubes or microtiter plates in a 5 x 5 grid format.
The Eppendorf Mastercycler® Personal is an essential instrument of our lab. For the last six years, it is being used for regular PCR of the DNA for cloning purpose. Apart from our lab, it is available to everyone in the institute for the use. The various reagents and buffers required for the PCR reactions have to be brought by the person using it.
Thermocycler Use in Life Science Research
Thermocyclers are integral to life science research. In molecular biology they are used for DNA sequencing, cloning, generation of probes, quantification of DNA and RNA, studying patterns of gene expression, detection of sequence-tagged sites, and many more techniques.

Thermocycler use extends beyond simple PCR and the amplification of nucleic acids to include both random and site-directed mutagenesis as well as the in vitro construction of recombinant DNA sequences. The fine temperature control and ability to hold a precisely set temperature with little fluctuation is also applicable to other techniques in which high-accuracy temperature control increases reproducibility between experiments. A thermocycler can be used in protocols where very strict temperature control is required such as heating tissue slides for in situ hybridization. The controlled temperature ramping provided by a thermocycler enables the study of temperature-dependent kinetics; for example, in addition to optimizing a PCR protocol, a thermocycler with a gradient feature allows for the determination of the optimum temperature for any enzymatic activity.
Thermocycler Use in Education
Several PCR machines and real-time PCR systems are available for educational use. All thermocyclers are fully functional standard machines. Using these PCR machines, students will gain a real research experience and an understanding of how PCR is being used in everyday testing.
Inquiry-based science teaching helps develop critical thinking skills. Integration of a memorable hands-on approach into the curriculum demonstrates practical applications of science, helps with retaining information, teaches how to interpret data, and keeps students engaged.
For life science education, a thermocycler is a good teaching tool for inquiry-based science. While learning how PCR analysis is used in practical ways, students are exposed to a range of scientific principles. Students can learn how PCR is used in fields such as forensics, diagnostics, animal conservation, and archaeology. A number of fun PCR amplification kits are available for teaching real-world PCR applications, for example, the Crime Scene Investigator PCR Basics™ kit and the Fish DNA Barcoding kit.
Thermocycler Use in Food Pathogen Detection
Speed is an important consideration for testing foods for pathogens. Many foodstuffs are perishable, so testing has to be fairly rapid or the food will not have a sufficient shelf life after testing is completed. For highly virulent pathogens such as E. coli O157:H7, speed and sensitivity is critical. This pathogen can be detected using a PCR kit in as little as 12 hours.
PCR is both more accurate and rapid than ELISA. Increased sensitivity means that an enrichment step is less likely to be required prior to assay. This shortens the overall time to getting a result and minimizes required hands-on time.
Use of automated PCR for pathogen testing, in addition to increasing speed, provides a higher throughput, lowers contamination, reduces the level of false positives, and decreases interlab variability. Walk-away automation provides automated DNA extraction and PCR setup, combined with automated data collection and analysis, bar codes for increased traceability, and LIMS integration.
Further, PCR kits are validated by several certification bodies. Validation studies have shown that the PCR kits are equivalent to, or better than, the various reference methods evaluated while providing results in much less time.
Culturing and ELISA were the main assays for testing for major food pathogens for many years. More recently, PCR assays and the ability to fully integrate a thermocycler into automated workflow have made pathogen testing faster and more sensitive, with a higher throughput.
Choosing a Thermocycler
There is an increasing array of types and uses of polymerase chain reaction, from reverse transcriptase PCR (RT-PCR) to Droplet Digital™ PCR. Which thermocycler, or thermocyclers, you choose depends on current and anticipated future needs. Considerations when choosing a thermocycler include the following:
- Types of PCR protocols used
- The level of sensitivity required
- The number of samples to be amplified at one time
- The range of sample volumes (analytical to preparative)
- Trade-offs between yield and specificity
- Whether gradient capability is required
- Protocol run times (e.g., for fast PCR)
- The number of users
One of the most difficult issues when buying a new PCR machine is deciding what your future needs will be. For instance, will there be a need for detection of low abundance targets? If so, Droplet Digital™ PCR might be a good option. If it is anticipated that there will be many different targets, a thermocycler with thermal gradient capability, providing rapid optimization of annealing temperatures, would be preferred
Six new thermocyclers were selected for this performance study: (A) Gene Amp 9700 (Applied Biosystems), (B) Multicycler PTC 200 (MJ Research, Inc.), (C) Tgradient (Whatman Biometra GmbH), (D) Mastercycler gradient (Eppendorf Netheler-Hinz GmbH), (E) Touchgene (Techne, Inc.), and (F) Primus 96 (MWG AG Biotech). All experiments were performed with the lid temperature set to 105 °C and with the maximum ramp rate available.
What are Thermocyclers?
Thermocyclers, also referred to as thermal cyclers, are heating cycler units that are utilized primarily for the amplification of RNA and DNA samples by a reaction known as polymerase chain reaction, or PCR. Thermocyclers achieve this through the process of raising and lowering the temperature of the samples, which are placed in a heating block platform. This heating and cooling process is regulated tightly and accurately to exact, pre-programmed parameters that define the temperatures used, the length of each cycle and the number of cycles. Via this process, nucleic acid samples are denatured and annealed within their individual sample tubes or wells with a mix of nucleic acids and reagents, including the enzyme polymerase. Amplifying nucleic acid is a prerequisite for most genetic applications and tests such as expression analysis, cloning, sequencing and genotyping of biological samples.
Different Types of Thermocyclers are Available
Standard Thermocyclers often are supplied with different block options, each of which can hold different formats of multi-well plates, tube strips or individual PCR tubes, and have a variety of setting and pre-programming options to meet various needs and applications. Real-time themocyclers, ideal for real-time, quantitative or "q" PCR, SNP analysis, drug target validation or genotyping.
Things to Think About Before Purchasing your Thermocycler
In order to identify the perfect Thermal cycler for your lab, you must first consider what you will be using the thermal cycler for. What sort of reactions and applications will you be using the unit for? Will it be primarily one reaction type of multiple different applications? What sort of throughput will you need? What sort of consumables will you need to use with your instrument? Will you require extremely high and accurate temperature ramp rates? Temperature gradient options are often available in Thermocyclers which allow the user to set up a range of temperatures across the one block in a particular program. This can be particularly handy when optimizing PCR reactions, such as optimizing primer annealing temperatures, for smaller or more valuable samples where you cannot afford to waste a single drop. You may also require space-saving Thermocyclers with a small footprint if you are short of space in your lab, or you may need a touch-screen interface, computer or wifi network connectivity, remote control of the unit, etc.